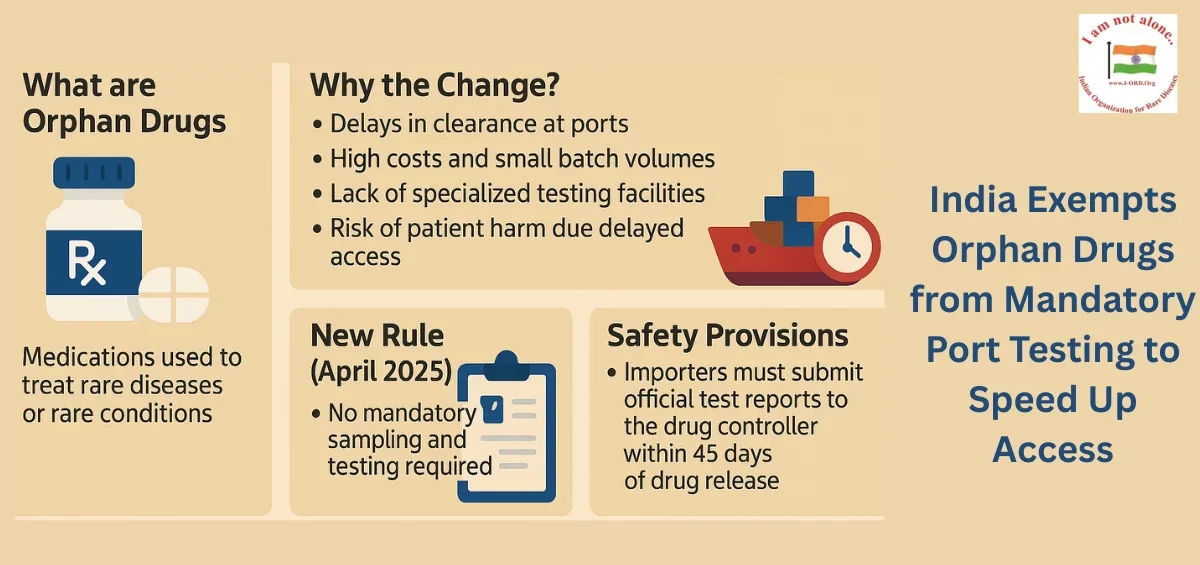
India waives port testing for orphan drugs, as announced by Central Drugs Standard Control Organisation, accelerating imports and improving timely access to life-saving treatments for rare disease patients.
Blog
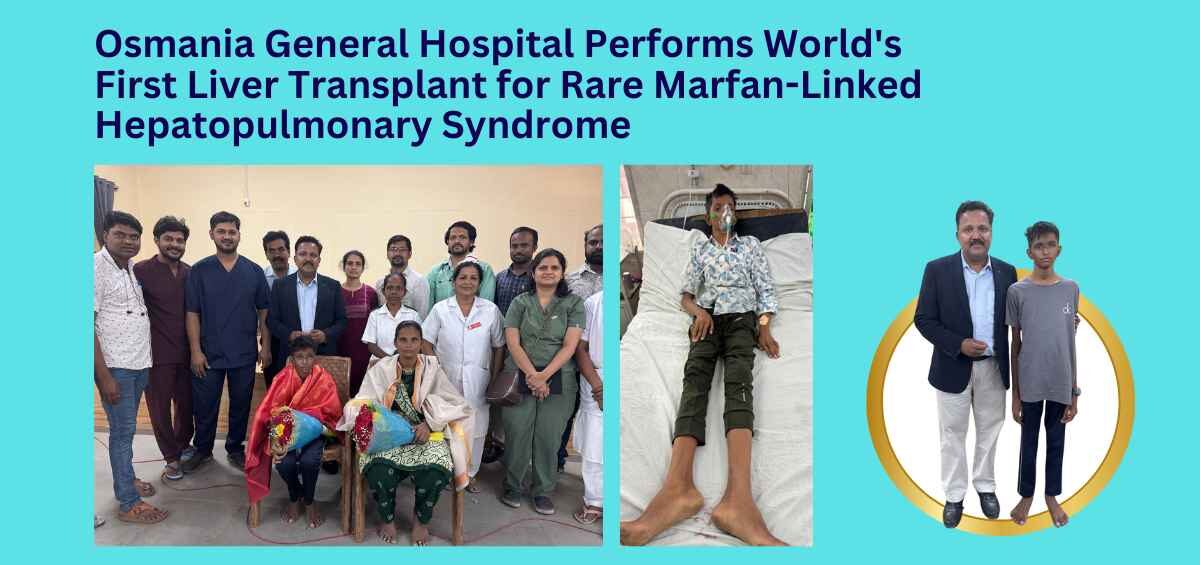
In a first, Osmania General Hospital doctors perform a life-saving liver transplant on a teen with Marfan Syndrome and severe Hepatopulmonary Syndrome.
IORD CEO and President Prof. Ramaiah Muthyala delivered keynote at the RJS PBH webinar on World Health Day 2025, emphasizing global action, early care, and hope for rare disease patients.